In a packed auditorium recently at New York University, some of the world’s most innovative neuroscientists and engineers were asked if they could predict the future.
“How are we going to unlock the mysteries of the brain and take that next step of discovery? What are we going to be talking about in the next decade?”
That was the question posed by Guy McKhann, MD, a neurosurgeon at Columbia University Medical Center and the moderator of an event organized as part of the 2017 World Science Festival. The event, a salon entitled “Engineering the Brain: Deploying A New Neural Toolkit,” brought together four researchers who, along with Dr. McKhann, shared their thoughts and their vision for how technology is transforming neuroscience.
Anthony Zador, PhD, Director of Science at Cold Spring Harbor Laboratories and a participant in this panel, argued that a key driver in making progress in neuroscience, a field that did not even exist 50 years ago, is the diversity of tools recently developed that can help scientists peer inside the brain with unprecedented clarity, whether they want to observe activity of a single neuron, or thousands of neurons working together that allow us to move, see or remember.
“We need to draw that line, from the activity of a neuron, through the connections between those neurons, to the ultimate behavior that results — that will give us the power to formulate the right next question,” he said.
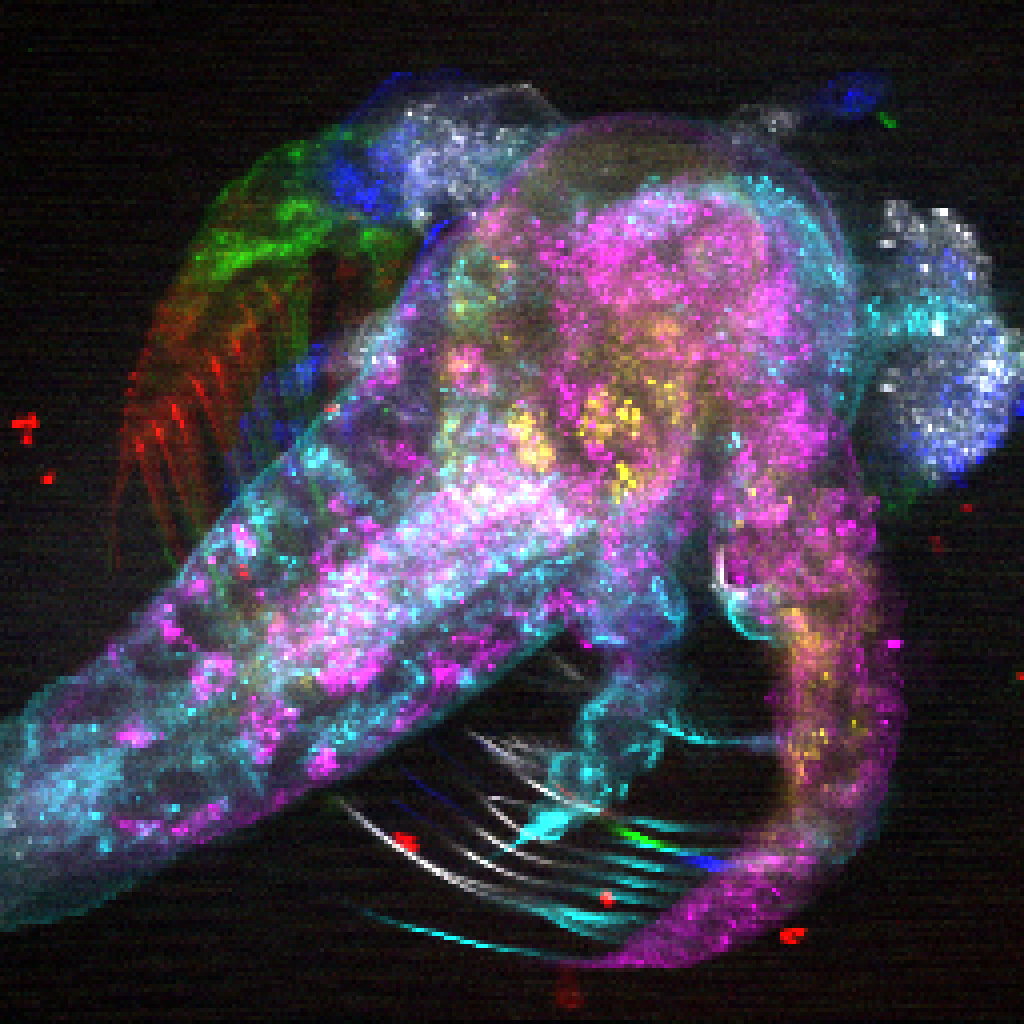
One of the most promising tools to be developed in recent years, and the panel’s first topic of discussion, was optogenetics — a technique that allows scientists to switch on individual brain cells, or groups of brain cells, in the brains of mice by using laser light.
For a long time, neuroscientists zoomed really far into the brain — a single cell at a time —to observe how one cell interacted with another. But when you’re able to zoom out, while still retaining that detail, it really changes the way that you think about the brain.
Panel participant Dayu Lin, PhD, a neurobiologist at New York University’s School of Medicine has used optogenetics in mice to study the brain-cell activity that underlies aggression and maternal instinct, two ancient behaviors that have persisted throughout the animal kingdom for hundreds of millions of years. Dr. Lin explained that when she started her career, optogenetics did not exist, leaving her to use electrical stimulation to turn on groups of neurons by sending an electrical pulse directly into the brain. But this technique had a downside: Electricity is difficult to control. Once the pulse passed into the neurons of interest, it was nearly impossible to prevent it from continuing its journey into neurons that were nearby.
“Optogenetics is advantageous because it allows you to really target the cells you’re interested in — and only those you’re interested in — without affecting neighboring cells,” said Dr. Lin. “It’s this specificity that makes it so powerful.” It was only with optogenetics that Dr. Lin could answer her questions about the brain cells that drive aggressive behavior.
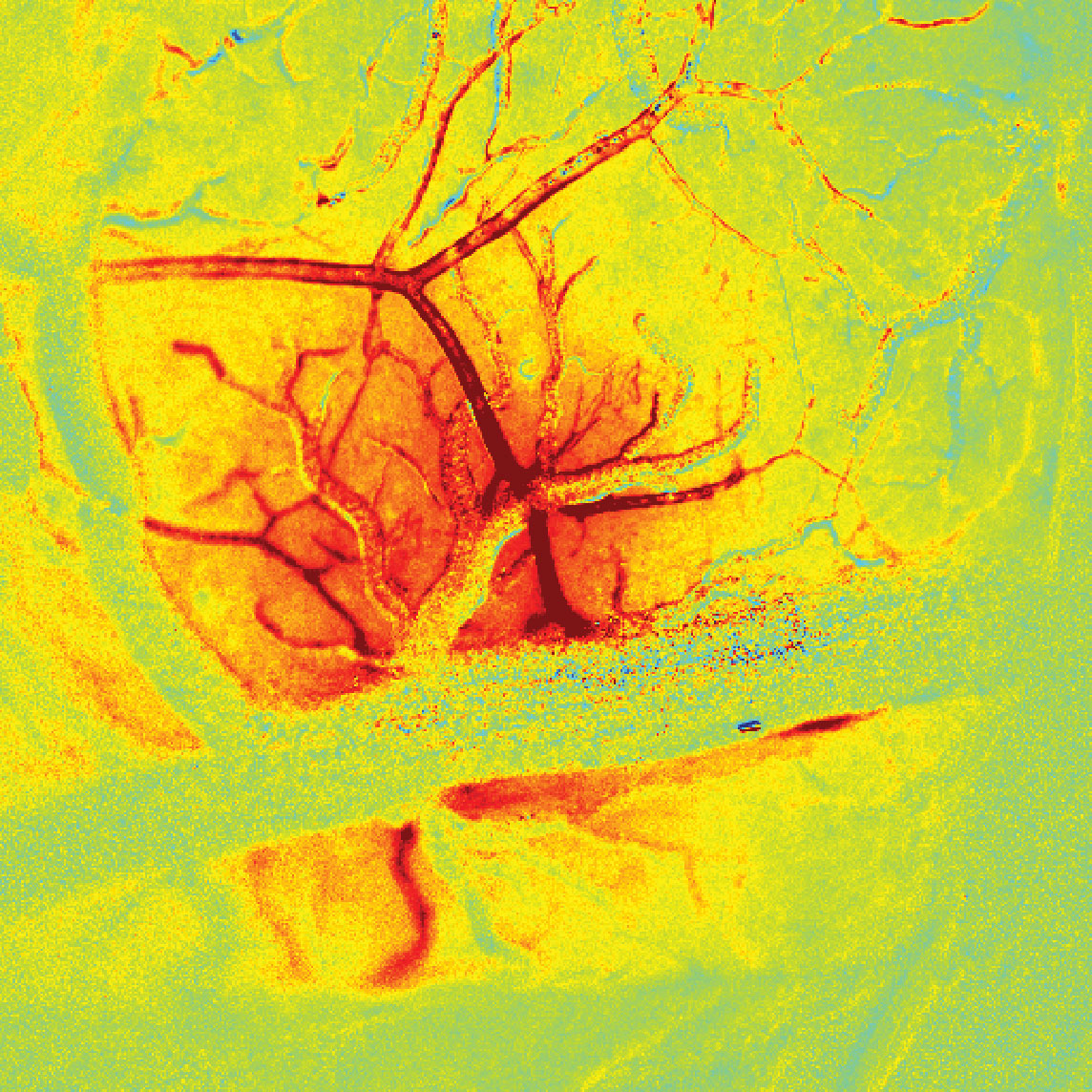
From manipulating cells with light to observing the whole brain in action, the conversation then pivoted to panel participant Elizabeth Hillman, PhD, a principal investigator at Columbia’s Mortimer B. Zuckerman Mind Brain Behavior Institute. One of Dr. Hillman’s main areas of interest is the development of new imaging methods that can capture the activity of individual neurons simultaneously across the entire brain. In one of her most recent studies, she and her team recorded the activity of neurons across the brain of a mouse while the brain was at rest. Their findings revealed that the brain was surprisingly active, even when it appeared to be doing nothing at all.
“For a long time, neuroscientists zoomed really far into the brain — a single cell at a time —to observe how one cell interacted with another,” said Dr. Hillman, who is also a professor of biomedical engineering at Columbia’s Fu Foundation School of Engineering and Applied Science. “But when you’re able to zoom out, while still retaining that detail, it really changes the way that you think about the brain.” This ‘new view’ of brain-wide neural activity could lead to a better understanding of how brain regions interact with each other, she said, and how these connections may be altered in disease.
The imaging tools that Dr. Hillman and her team use in her lab are both powerful and precise, but have been designed with smaller species in mind, such as mice and fruit flies. There is no equivalent tool for humans, though there is something similar, called functional magnetic resonance imaging, or fMRI. This technique works similarly to a regular MRI machine, but by detecting small changes in images that occur when a person performs tasks, such as squeezing a ball or playing a video game, fMRI can help determine how different brain regions are involved in everything from learning a new skill to recalling a memory.
But there’s a significant problem when it comes to fMRI: it isn’t actually measuring brain activity. Rather, it traces changes in blood flow as a proxy for brain activity. As a result, the technique has come under fire by those skeptical of how accurately it can be used to represent brain activity. This relationship between blood flow changes and neural activity is another area that Hillman studies in her laboratory.
“fMRI has a bit of an image problem,” said Dr. Hillman, “But getting a better handle on the relationship between blood flow and brain activity will give us a better way to use fMRI, expanding what we can learn about the human brain.”
Conor Liston, PhD, a neuroscientist at Weill Cornell Medicine and the panel’s fourth participant, agreed that fMRI has massive potential as a tool for studying the human brain — especially if it can be used effectively to understand disease.
In Dr. Liston’s work on psychiatric disorders such as depression, he hopes that an increased understanding of the underlying mechanisms of the disorder — as viewed through fMRI — could lead to more specific diagnoses and, ultimately, more effective treatments.
“We want to figure out whether we can identify clusters of patients that have similar biological features, as indexed by fMRI,” he said. “And if so, then maybe we can develop specific treatments targeted to those patients who are most likely to benefit from them.”
Throughout the 90-minute discussion, a common thread that arose was the importance of diversity in the field of neuroscience. The questions addressed by scientists who study the brain are so vast, and so complex, the panel agreed, that it will take experts from a great many of fields — and with a variety of insights — to answer them.
“Science needs lots of different viewpoints,” said Dr. Hillman. “All of us on this stage have in our labs students from around the world, of every race, gender and background. They all bring completely different perspectives, and it is that richness that moves science forward.”