Survival might not be top of mind when you are feasting on a filet mignon in butter sauce on a night out, but the taste receptors sparked into action with each bite are, evolutionarily speaking, what draws you to foods that keep you nourished. Similarly, the revulsion you feel from a bite of anything too bitter, sour, or salty is what keeps you from ingesting a poisonous substance that could kill you.
Taste has two purposes, proposed the gastronome Jean Anthelme Brillat-Savrin in his 1826 treatise, “The Physiology of Taste.” “It invites us, by arousing our pleasure, to repair the constant losses which we suffer through our physical existence,” he writes, and “it helps us to choose from the variety of substances which Nature presents to us those that are best adapted to nourish us.” And yet, despite how clear are our experiences of taste, he notes, “It is not easy to determine precisely what parts make up the organ of taste. It is more complicated than it seems.”
Scientists who have studied the question in the nearly two centuries since then would undoubtedly agree with this assessment. Taste was the last of the five senses to give up the broad brushstrokes of its molecular secrets. In the past 15 years or so, a coherent picture of taste perception has emerged, in large part through the work of Charles Zuker, PhD, professor of biochemistry & molecular biophysics and of neuroscience at P&S since 2009. During that time, Dr. Zuker and his colleagues elucidated the basic organization of taste perception in the tongue, pinning down one by one the identity of all five types of taste receptors and cells: sweet, bitter, sour, salty, and umami. More recently, they also established the basics of how signals transmitted through these receptors are interpreted in the brain.
“Zuker’s work really provided the most significant insight and knowledge about the sense of taste,” says Richard Axel, MD, University Professor and co-recipient of the 2004 Nobel Prize in Physiology or Medicine for his work identifying the genes that encode olfactory receptors. “The thing about Charles is that he does science with real creativity and courage. He goes in and breaks open a difficult problem.”
I realized that the fundamental premises that were at the time the de facto understanding of how taste operated made no sense.
Dr. Zuker began his career in the mid-1980s investigating the molecular signaling pathway between the retina and the brain in flies, but in the late 1990s he set his scope on taste, where it has remained ever since. The choice of sensory system really doesn’t matter, he says; the greater goal is to figure out how the brain translates sensory cues it receives from the external world into meaningful signals that make an organism tick. “The beauty about taste is that it has limited chemical space. There are only five basic qualities and each of them has an innate and very well-defined meaning and valence,” he says. “So the taste system provides a powerful platform to understand how the brain functions: how complex, hard-wired circuits operate, how they are modulated, and how they are used to guide actions and behaviors.”
Finding the Right Experiment
When he speaks about his work—hands gesturing expansively to punctuate descriptions of experiences and experiments, glasses perched atop his head—Dr. Zuker exudes intensity as well as a kind of laser-like clarity and devotion to logic. He seems guided by the idea that the world breaks down into propositions that will stand or fall on the outcome of an experiment—as long as you can find the right one. It is a mindset that he adopted early in life: He has been pretty much committed to uncovering the secrets of the natural world since receiving a microscope as a gift from his parents when he was 7 or 8 years old.
After studying biology in college in his native Chile, Charles Zuker headed to the Massachusetts Institute of Technology for graduate school at the age of 19. He soon found his way to the lab of cellular and developmental biologist Harvey Lodish, who was studying gene regulation and differentiation of the cellular slime mold Dictyostelium discoideum. Dr. Zuker’s flair as a researcher was unforgettable, says Dr. Lodish. “He did quite an exceptional piece of work in my lab. It was some of the first cloning of developmentally regulated genes in the organism. He was brilliant, brash, arrogant, very talented, very productive.” At one point, Dr. Zuker declared his research completed and assembled a thesis committee to evaluate him; it turned out, however, that committee members disagreed and he returned to the bench for another year of experiments. The hiccup changed the course of his future work. During that year, Dr. Zuker’s plans to pursue a postdoc in a high profile European cloning lab crumbled and his interest in studying the neurobiology of complex organisms crystallized. In 1983 he joined the lab of one of Dr. Lodish’s earlier mentees, Gerald Rubin, at the University of California, Berkeley.
At the time, researchers were just beginning to develop the tools to probe the nervous system at the level of molecular biology, and Dr. Rubin (now executive director of the Howard Hughes Medical Institute’s Janelia Research Campus) was at the forefront of this effort. The previous year, the Rubin lab had invented a method to alter the fly genome by inserting bits of DNA into specified locations, allowing researchers to probe the function of genes by mutating them. “This was a transforming, disruptive technology that changed what you could do with flies, because now, for the first time, you could examine causality by manipulating it,” Dr. Zuker says. In the four years he spent in Dr. Rubin’s lab, he helped identify the fly genes encoding different forms of rhodopsin, a light-sensitive protein in the retina whose activation kicks off photoreception, and probed the genes’ function by mutating and manipulating them in living flies.
A Surprising Turn to Taste Research
In 1987 Dr. Zuker started his own lab at the University of California, San Diego, where he went on to dissect the phototransduction pathway in flies, using genetics and physiology to uncover the underlying signaling mechanisms and using it as an entry point to understand how G-protein coupled receptors (GPCRs) can transform a photon of light into a signal that activates a photoreceptor neuron. With this focus on signal transduction in the visual system, the lab’s foray into taste was something of a fluke. It began with a commentary that Dr. Zuker was asked to write in 1995 for a paper published in Nature on GPCR signaling in taste transduction. Very little was known about the molecular mechanism underlying taste and, despite decades of searching, researchers had never been able to identify taste receptors. The more Dr. Zuker read about the field, the more perplexed he became. “I realized that the fundamental premises that were at the time the de facto understanding of how taste operated made no sense,” he says.
For one thing, the widely held view was that taste receptor cells were broadly tuned for the five known taste qualities and that the brain would later deconvolute this complex signal. “We figured that it’s inconceivable that you would mediate something as fundamental to life and death by using a system that relies on broadly tuned sensors,” he explains. Another thing that bugged him was that researchers had proposed a mishmash of different types of receptor signaling systems for mediating different taste qualities. “The nature of the receptors that had been proposed was not consistent with what we knew about sensory receptors.”
His lab had always included a few researchers whose work lies outside of the group’s core interests, so he set a new postdoc to work on the topic. He also struck up a partnership with Nicholas Ryba, a geneticist studying taste and smell at the National Institute of Dental and Craniofacial Research. Dr. Ryba similarly smelled something fishy in the taste field’s status quo. The two teams began a concerted search for taste receptors by screening RNA sequences in taste buds from the rodent tongue, and in 1999 they reported the identification of two novel GPCRs, T1R1 and T1R2, that seemed to fit the bill.
But it took a larger resource—specifically, the draft sequence of the human genome, which was published the following year—for their efforts to really hit pay dirt. Earlier genetic studies had pinpointed a gene on chromosome 5 that was associated with the ability to taste a bitter substance called 6-n-propyl-2-thiouracil, or PROP, so the duo combed the draft sequence for orphan GPCRs and identified an entire family of bitter-sensing GPCRs. These so-called T2R receptors were present in varying combinations in one type of taste receptor cell, and the combination would determine a person’s sensitivity to bitter taste.
The work, published in June 2000 as a pair of studies in the journal Cell, was one of the first discoveries to be made by mining the human genome. It also spurred a race among taste researchers in the search for other types of receptors. The following year, Drs. Zuker and Ryba identified a third T1R, T1R3, and explained the role this family of receptors plays in taste. These receptors’ pairing in a cell determined its function, it turned out: T1R2 and T1R3 combined for detecting sweet, while T1R1 and T1R3 together sensed umami, a savory taste that in humans is elicited by the amino acid glutamate. The findings stacked up to build a picture of different taste qualities encoded by distinct receptors expressed on dedicated cells. “At the time, these things were such exciting discoveries; they ran so counter to the dogma,” says Dr. Ryba.
Studying taste receptors soon went from being a side project to the primary interest of Dr. Zuker’s lab. Progress was steady, as a regular stream of high-profile papers published exclusively in Cell, Nature, and Science attest. In addition to finding sweet, bitter, and umami receptors, the Zuker and Ryba teams found the receptors and taste cells activated by sour and salty stimuli as well as the sensor for carbonation. To define how each receptor system worked, they knocked out the gene for all the receptor types in mice, thereby selectively abolishing the ability of the mutant animals to perceive the corresponding taste. “The fact that other tastes remain unaffected shows something very important about taste coding,” says Dr. Zuker. “It shows that each taste is mediated by receptors and pathways independent of every other.” Conversely, they could expand the animals’ taste repertoire by introducing receptors that are usually absent. For example, mice are insensitive to many artificial sweeteners, including aspartame. “So we cloned the human receptors and we did the obvious experiment, inserting that human receptor into mice. And indeed, we completely humanized their sweet taste responses.”
A Switch to Mouse Genetics
Perhaps the biggest challenge early on, Dr. Zuker says, was the patience required in switching from fly to mouse genetics. As molecular biology techniques, such as the ability to knock out specific genes in the mouse, continued to advance, working in mammalian systems was the obvious choice. However, if you want to tweak the genome of a fruitfly, you can have a mutant line up and running within a few months. Engineering a mouse from the beginning, on the other hand, could take more than a year. The researchers also had to devise a whole set of behavioral paradigms to test the animals’ responses. And of course, they encountered the occasional conceptual blind alleys. “For the longest time, for example, we believed there must be a sixth category of taste receptor cells that would respond to high salt,” says Dr. Ryba, since this taste is strongly aversive. In 2013 they discovered that this taste quality is encoded differently: Rather than having a separate set of receptors, salt at high concentrations causes aversion by activating bitter and sour receptors.
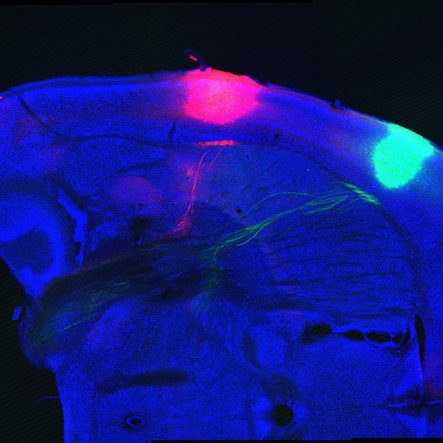
The most difficult scientific frontier, however, turned out to be connecting how these signals from the periphery were processed in the brain. “From the get-go, the goal was to use the receptors to follow the signal and ultimately understand how the brain knows what the tongue knows—how the brain represents the world,” says Dr. Zuker. Different taste qualities are reliably distinct, he and Dr. Ryba reasoned, so their representation in the cerebral cortex must be as well. But tracing taste receptor neurons that project to the brain turned out to be technically tough, and the key brain area could not be accessed without some extra finagling. It took until 2011 to get the first snapshot of taste representation in the brain, but the results were worth the wait: All the taste qualities segregated into their own, distinct region of the taste cortex in mice, forming a gustatory map. The fact that this delineation was so clear “was a complete surprise,” says Dr. Ryba, “but it makes a lot of sense.” It allows each type of taste receptor to be wired independently, perhaps stemming from evolutionary developments of different tastes. In a more recent study, published last November, Robert Barretto, a postdoc in Dr. Zuker’s lab, showed that neurons relaying the signal from taste receptors to the brain are also tuned to a single taste quality, suggesting that information from the tongue essentially travels along dedicated, labeled lines all the way to the brain.
Dr. Zuker’s lab research jives closely with a small core of other molecular neuroscience labs throughout Columbia that investigate the molecular dimension of sensory systems. For example, Richard Axel, who laid the original groundwork for understanding how the brain interprets smell by identifying olfactory receptors, currently studies how complex organisms develop learned responses to odors and how learning and experience give these odors meaning. “Sort of like the smell of madeleines for Proust summon forth his childhood,” Dr. Axel says. The work of Tom Maniatis, chair of biochemistry & molecular biophysics, on proteins called protocadherins, which play an important role in determining neural circuitry, overlaps with both Dr. Zuker’s and Dr. Axel’s work. “We all see each other every day and talk about our science,” Dr. Maniatis says. Another scientist working on the sense of smell, Stavros Lomvardas, PhD, brought his lab to Columbia from the University of California, San Francisco, in September. While Dr. Zuker and Dr. Axel focus on how the brain interprets taste and smell, Dr. Lomvardas, who did his PhD and postdoc training at Columbia, studies the process by which olfactory neurons at the periphery manage to express any one particular smell receptor out of the enormous diversity of those encoded in the genome.
Further exploring how information from taste receptors in the tongue is interpreted and organized by the brain continues to be the main project of Dr. Zuker’s lab. The lab also has begun to explore the interplay between taste and other senses and higher order cognition like learning and memory. “The brain is this chunk of tissue that is made out of a hundred billion neurons that transforms the human condition. It changes fear into courage, sadness into happiness, conformity into creativity,” says Dr. Zuker. “What is the substrate for creativity in the brain? I don’t know. But studying the senses gives us a magical window into the workings of the brain.”
This article originally appeared in Columbia Medicine magazine.