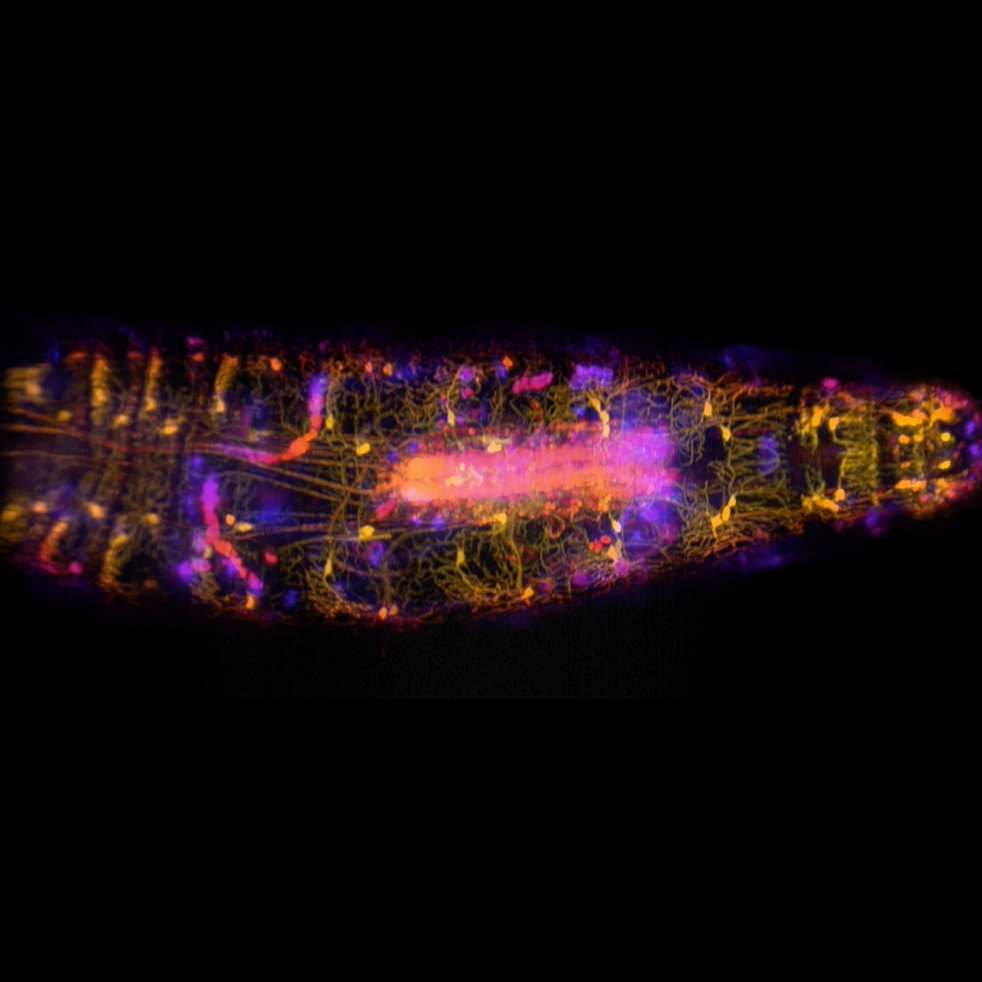
The Columbia team behind the revolutionary 3D SCAPE microscope announces today a new version of this high-speed imaging technology. In collaboration with scientists from around the world, they used SCAPE 2.0 to reveal previously unseen details of living creatures — from neurons firing inside a wriggling worm to the 3D dynamics of the beating heart of a fish embryo, with far superior resolution and at speeds up to 30 times faster than their original demonstration.
These improvements to SCAPE, published today in Nature Methods, promise to impact fields as wide ranging as genetics, cardiology and neuroscience.
Why is having faster, 3D imaging so valuable? “The processes that drive living things are dynamic and ever-changing, from the way an animal’s cells communicate with one another, to how a creature moves and changes shape,” said Elizabeth Hillman, PhD, a principal investigator at Columbia’s Mortimer B. Zuckerman Mind Brain Behavior Institute and the paper’s senior author. “The faster we can image, the more of these processes we can see — and imaging fast in 3D lets us see the whole biological system, rather than just a single plane, offering a clear advantage over traditional microscopes.”
SCAPE 2.0 opens up a new landscape of things that we can see. I hope our new results will inspire scientists to think of what new questions can be asked, and what new avenues of scientific discovery we can explore next.
When Dr. Hillman’s team first introduced SCAPE (swept confocally aligned planar excitation) microscopy four years ago, their approach challenged assumptions about how to create an image of living tissues at high speeds.
“Most microscopes that image living samples scan a small spot of laser light around the sample, but the point-scanning approach is slow, giving only a short time to see each spot,” said Venkatakaushik Voleti, PhD, the paper’s first author who developed SCAPE 2.0 as a doctoral candidate in Dr. Hillman’s lab. “Our system uses an oblique, or angled, sheet of light to illuminate an entire plane within the sample, and then sweeps this light sheet across the sample to form a 3D image.”
Although imaging samples using sheets of light date back more 100 years, SCAPE’s ingenuity lies in the way that it rapidly moves the light sheet and focuses the image of this sheet back to a stationary camera using a single moving mirror — making it lightning fast and surprisingly simple. In addition, SCAPE is gentle on living samples because it uses only a fraction of the light that point-scanning microscopes would need to get images at comparable speeds. SCAPE achieves all this through a single, stationary objective lens, opening up space for a wide array of samples compared to conventional light-sheet microscopes that require complex sample chambers surrounded by many lenses.
“People are often surprised at how compact, simple and easy to use SCAPE is,” said Dr. Hillman, who routinely drives SCAPE systems around in the trunk of her car to give researchers hands-on demonstrations.
Dr. Hillman’s team is working to help scientists all over the world use SCAPE for their own research, inviting scientists to her lab at Columbia’s Zuckerman Institute, or helping them to build their own systems, thanks to grant support from the National Institutes of Health BRAIN Initiative. Dr. Hillman is also working with Leica Microsystems, who have licensed SCAPE and are currently developing a commercial version of the system.
Dr. Hillman attributes broad interest in SCAPE 2.0 to recent major advances in fluorescent labeling, which lets scientists make specific cells in an animal glow different colors, and can even make cells flash on and off when they are signaling to each other. She also notes the growing impact of small, near-transparent animals such a C. elegans worms, zebrafish embryos and fruit flies which can be observed during natural behaviors, or be modified to recapitulate human diseases. SCAPE 2.0 is perfectly positioned to capture the symphony of cellular events, movements and responses playing out in these living systems.
“In our new paper, we show how SCAPE 2.0 can track individual neurons firing in a whole animal as it crawls around, giving us a new window into how neural activity guides behavior,” said Dr. Hillman, who is also professor of biomedical engineering at Columbia Engineering.
Despite being inspired by neuroscience needs, Dr. Hillman notes that many of the aforementioned labeling methods and animal models are now transforming other research areas, letting scientists explore how cancerous tumor cells signal to each other, how immune cells find their targets or how the heart and cardiovascular system are affected by drugs and disease.
“It is really exciting to see techniques, stimulated by the BRAIN initiative, having ever broader impacts on science and medicine” said Dr. Hillman.
Recognizing this opportunity, Dr. Hillman partnered with pediatric cardiologist Kimara Targoff, MD, to put SCAPE 2.0 to work in studying how the heart develops. Dr. Targoff’s lab uses zebrafish as an animal model to decipher the genetic mutations that can cause heart malformations in the embryo. Understanding how these mutations lead to disease could inform treatments for children living with congenital heart disease.
“The problem with imaging the beating heart is that it beats fast, changing its shape as blood flows through it in a wide range of directions,” said Dr. Targoff. who is an assistant professor of pediatrics at Columbia’s Vagelos College of Physicians and Surgeons and a co-author of today’s paper. “With SCAPE 2.0, we can image the zebrafish embryo’s beating heart in 3D and in real-time, allowing us to see how calcium signals sent between heart cells cause the heart wall to contract, or how red blood cells flow through the heart’s valves beat after beat. Using this knowledge, we can track how a particular genetic mutation affects normal heart development in an environment that most closely recapitulates the heart’s natural state.”
The desire to follow a single red blood cell as it travels through the beating heart was a driving force behind pushing the speed limits of SCAPE 2.0.
To reach these unprecedented speeds, Dr. Hillman’s team worked closely with Lambert Instruments, leveraging the company’s ultra-fast HiCAM Fluo camera. This camera was used to capture images at more than 18,000 frames per second in the zebrafish embryo’s beating heart. This new configuration opened the door to recording individual neurons firing in a freely moving C. elegans worms, giving the first view of an animal’s complete nervous system in action. SCAPE 2.0’s other upgrades include improved light efficiency, a larger field of view and much improved spatial resolution.
SCAPE 2.0 image of an mCubic cleared mouse brain (Thy1-GFP). The 8.4 x 9 x 0.4 mm xyz volume was acquired within 4 minutes with 1 x 1.37 x 1.14 micron per pixel sampling density. Rendered using Imaris (Bitplane). Sample provided by Pavel Osten Lab (Credit: Kripa Patel/Hillman lab/Columbia’s Zuckerman Institute).
SCAPE 2.0’s improved resolution also enabled the team to image samples created using tissue clearing and tissue expansion. These methods let scientists see structures and connections deep inside intact samples, from whole mouse brains to tumors and human biopsies. Although these samples are not alive, they are very large and take a long time to image using standard microscopes. Today’s paper demonstrates that SCAPE 2.0 could image these types of samples at record-breaking speeds
Dr. Hillman and her team are continuing to develop and improve SCAPE to further expand its utility, while working with an ever-growing group of collaborators, from Zuckerman Institute neuroscientists to Columbia volcanologist Einat Lev PhD, who is using SCAPE to image the way gas bubbles form during volcanic eruptions.
Dr. Hillman’s team is also developing a miniaturized version of SCAPE for medical use, to quickly distinguish between healthy and diseased cells within a patient’s body, giving doctors a new way to guide how to perform complex surgeries in the operating room.
“The limitations of tools and techniques often constrain what scientists think they can study,” said Dr. Hillman, who is also professor of radiology at Columbia’s Vagelos College of Physicians and Surgeons. “SCAPE 2.0 opens up a new landscape of things that we can see. I hope our new results will inspire scientists to think of what new questions can be asked, and what new avenues of scientific discovery we can explore next.”
###
This paper is titled "Real-time volumetric microscopy of in-vivo dynamics and large-scale samples with SCAPE 2.0" (open access link to download). Additional contributors include Kripa Patel, Wenze Li, PhD, Citlali Campos, Srinidhi Bharadwaj, Hang Yu, Caitlin Ford, Malte Casper, Richard Wenwei Yan, Wenxuan Liang, PhD, Chentao Wen, PhD, and Koutarou Kimura, PhD.
This research was supported by the National Institutes of Health BRAIN Initiative (5U01NS09429, UF1NS108213, U19NS104649, R01HL13143801A1), the National Science Foundation (NSFGRFP, IGERT, CAREER CBET-0954796), the Simons Foundation Collaboration on the Global Brain (542951), Department of Defense (MURI W911NF-12-1-0594), the Kavli Institute for Brain Science, and the Columbia-Coulter Translational Research Partnership and Coulter Foundation.
SCAPE intellectual property is licensed to Leica Microsystems for commercial development. Venkatakaushik Voleti, Wenze Li, Kripa Patel, Citlali Campos and Elizabeth Hillman declare a financial interest in SCAPE microscopy via consulting activity and a license agreement in place between Columbia University and Leica Microsystems for commercial development of SCAPE.