When Stavros Lomvardas, PhD, was a graduate student, he did not set out to study the brain. He was fascinated by an even more fundamental aspect of biology: what makes one cell different from the next.
This fascination led him to study the class of brain cells that give us our sense of smell, olfactory neurons. He investigates the genes that generate different types of olfactory neurons and give us the power to parse billions of different scents.
In just a few years, Dr. Lomvardas’ research has already provided new insights into the organization and structure of our olfactory system. His work has also caught the attention of the Howard Hughes Medical Institute (HHMI), which today announced that Dr. Lomvardas is among the newest members of their Faculty Scholars Program.
The HHMI Faculty Scholars program recognizes accomplished, early career researchers with strong potential to make groundbreaking contributions to science and medicine. He will receive $900,000 over a period of five years. We spoke with Dr. Lomvardas, a principal investigator at Columbia’s Mortimer B. Zuckerman Mind Brain Behavior Institute and professor of biochemistry and molecular biophysics at Columbia University Medical Center (CUMC), about his work and how this recognition from the HHMI will support his research about the science of smell.
Your early work focused on the study of genetics. How did that lead to your interest in the science of olfaction?
Originally, I wanted to understand the biological steps that generate the vast array of cells types in the body. The best way to do this is to look at the genetic material inside each cell. Almost every cell in your body has the exact same set of genetic material, collectively known as our genome. But it is the pattern of which genes get switched on and which remain dormant that makes one cell different from the next.
Under the mentorship of Dr. Richard Axel, a Zuckerman Institute codirector who won the 2004 Nobel Prize for his studies on olfaction, I became interested in how gene regulation operates in the olfactory system. And the closer I looked, the more complex and mind-boggling it began to appear.
What was most mind-boggling about the olfactory system?
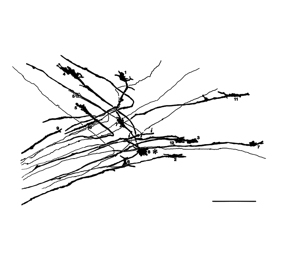
There are about 1000 genes involved in olfaction. In almost all cases, specialized proteins called transcription factors activate the right gene at the right time by binding directly to it. But genes in olfactory neurons are packed into an unusually dense, compact bundle. We had trouble figuring out how any transcription factor could wrest away a single gene from that bundle in order to activate it.
Early on, Dr. Axel and I hypothesized that a single, specialized structure was guiding this process, though at the time we had no way of knowing for sure. But then a few years ago, when I was a principal investigator at the University of California, San Francisco, my collaborators and I found evidence supporting this idea.
In research published in Cell in 2014, we identified that complex, three-dimensional structure. Somehow this structure allows for a single olfactory gene to pop out of the tightly packed cluster and become activated. Precisely how this occurs is something we’re now trying to figure out.
What are the long-term implications of this research?
Our studies on gene regulation of olfaction could have profound implications for other areas of the nervous system. There are many gene families involved in the nervous system that remain largely uncharacterized. If we can uncover more about how genes in different systems are physically organized — how they cluster together, how they become activated — we’ll get a much clearer window into the underlying principles of the brain.
Importantly, our work is also shedding light on what happens when this organizational structure goes awry, for example in various disorders where neurons fail to develop properly. Moreover, some of the regulatory principles we are uncovering may shed light on conditions that on the surface appear to be completely unrelated to our research, for example in Alzheimer’s disease.
What discoveries related to Alzheimer’s have you made so far?
For a long time, there was anecdotal evidence that a decreased ability to smell was an early indicator of Alzheimer’s disease. That connection was largely ignored, but recent research performed by Dr. Devangere Devanand and colleagues at Columbia University Medical Center provides strong correlations in humans between deficits in olfaction and Alzheimer’s. Intriguingly our work suggests that olfactory neurons have some unique characteristics that may support this link.
Almost every type of cell, including the vast majority of neurons, contains proteins that act as a quality control system, a sort of clean-up crew, that finds damaged or misfolded proteins and flushes them away. Intriguingly, olfactory neurons have no such quality control system, so misfolded proteins tend to accumulate at higher levels than in other types of neurons.
Alzheimer’s disease is, ultimately, caused by an abnormal buildup of proteins in neurons, which leads to these cells’ degradation and death. Our current hypothesis is that because olfactory neurons lack this cellular clean-up crew, they are the part of the nervous system first affected by Alzheimer’s, potentially explaining why anosmia (a loss of the sense of smell) may be early predictors of this disease.
How will becoming an HHMI Faculty Scholar affect your research programs?
One of HHMI’s guiding principles is to support people, not projects. Their backing will give me the freedom to follow ideas, to follow the science, wherever it may lead. In our lab, we have many ideas that we cannot yet explore because we do not have funding. With HHMI’s support, we will be able to take risks and test unconventional ideas that may yield groundbreaking results.