NEW YORK — For millions of cancer patients, life-saving chemotherapy comes at a cost: relentless, chronic nerve pain, numbness or tingling emanating from the fingers and toes that often emerges during treatment. This condition, called chemotherapy-induced peripheral neuropathy (CIPN), can be set off by even the lightest touch. There are no effective treatments.
But a new Columbia study in flies and mouse cells has revealed a promising research direction for curbing this disorder: a protein called integrin that can potentially curb CIPN by protecting nerve cells against damage.
These findings were reported today in Proceedings of the National Academy of Sciences.
“Chemotherapy drugs can successfully bring patients into remission, but these drugs are often accompanied by unbearable nerve pain,” said Wesley Grueber, PhD, a principal investigator at Columbia’s Mortimer B. Zuckerman Mind Brain Behavior Institute and the paper’s senior author. “Treatment for this pain can consist of a combination of therapies, including pain management with analgesics including opioids, antidepressants or anti-seizure drugs, or halting chemotherapy until symptoms improve — a strategy that may endanger the life of the patient. New strategies for treating, and preventing, CIPN are badly needed.”
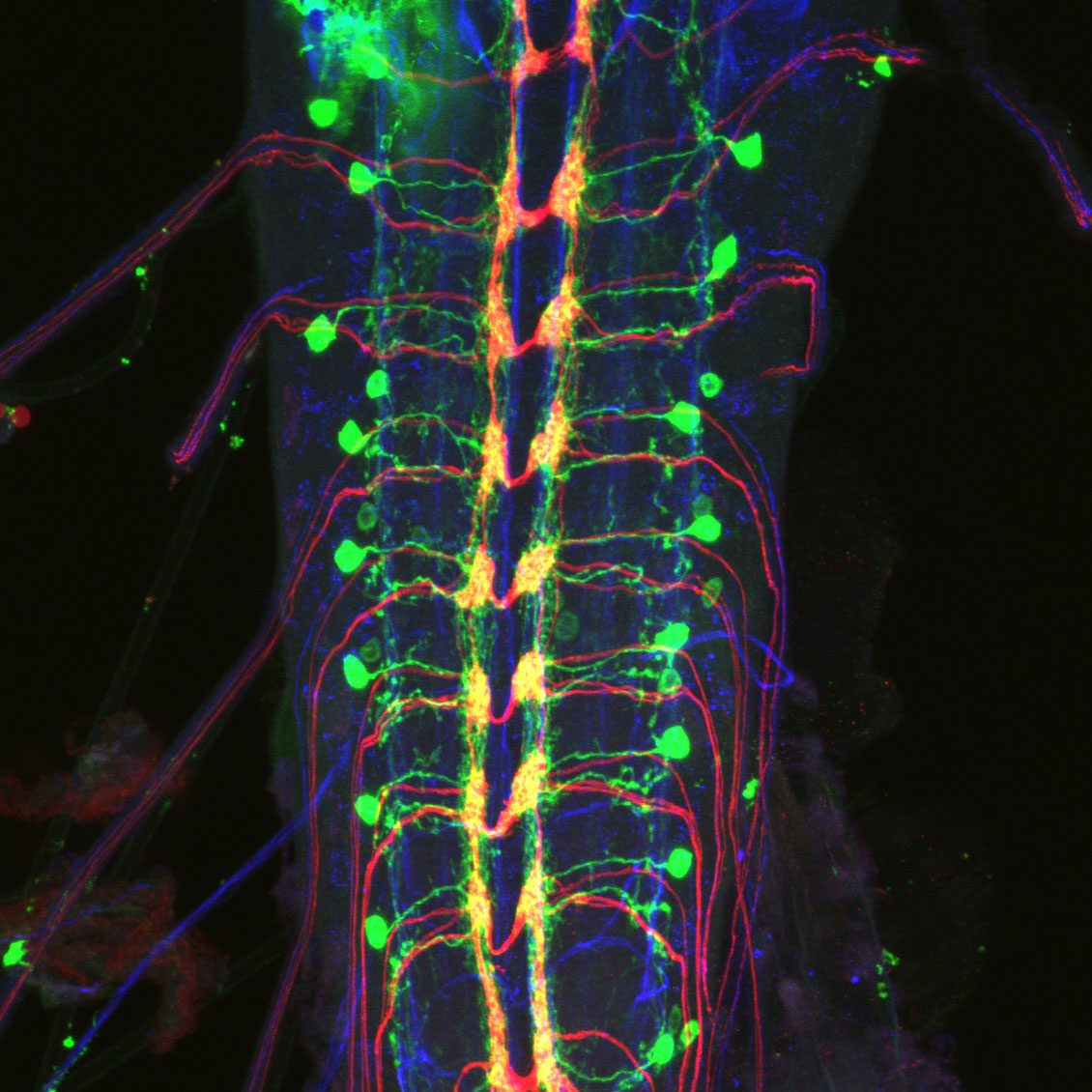
Our sense of touch and pain is extraordinarily sensitive, thanks to nerve-cell endings that extend like branches from our spinal column to our fingertips. These branches are essential for performing dexterous movements, like playing a violin. They also alert us to dangers, such as extreme heat or sharp objects. Typically, these nerve-cell branches are organized in such a way to completely cover the skin. Our bodies have evolved ingenious methods of maintaining this arrangement.
“Nerve endings are organized by the ecosystem that surrounds them, a kind of microscopic scaffolding dotted with adhesion points that effectively glue cells in the right places and keep them healthy,” said Grace Shin, PhD, an associate research scientist in the Grueber lab and the paper’s first author. “Integrins are proteins that play an essential role in this adhesion process.”
In 2012, Dr. Grueber and his team found that integrins spur the healthy growth of developing nerve cells. Integrins also guide nerve cell repair, especially at the body’s peripheral nervous system.
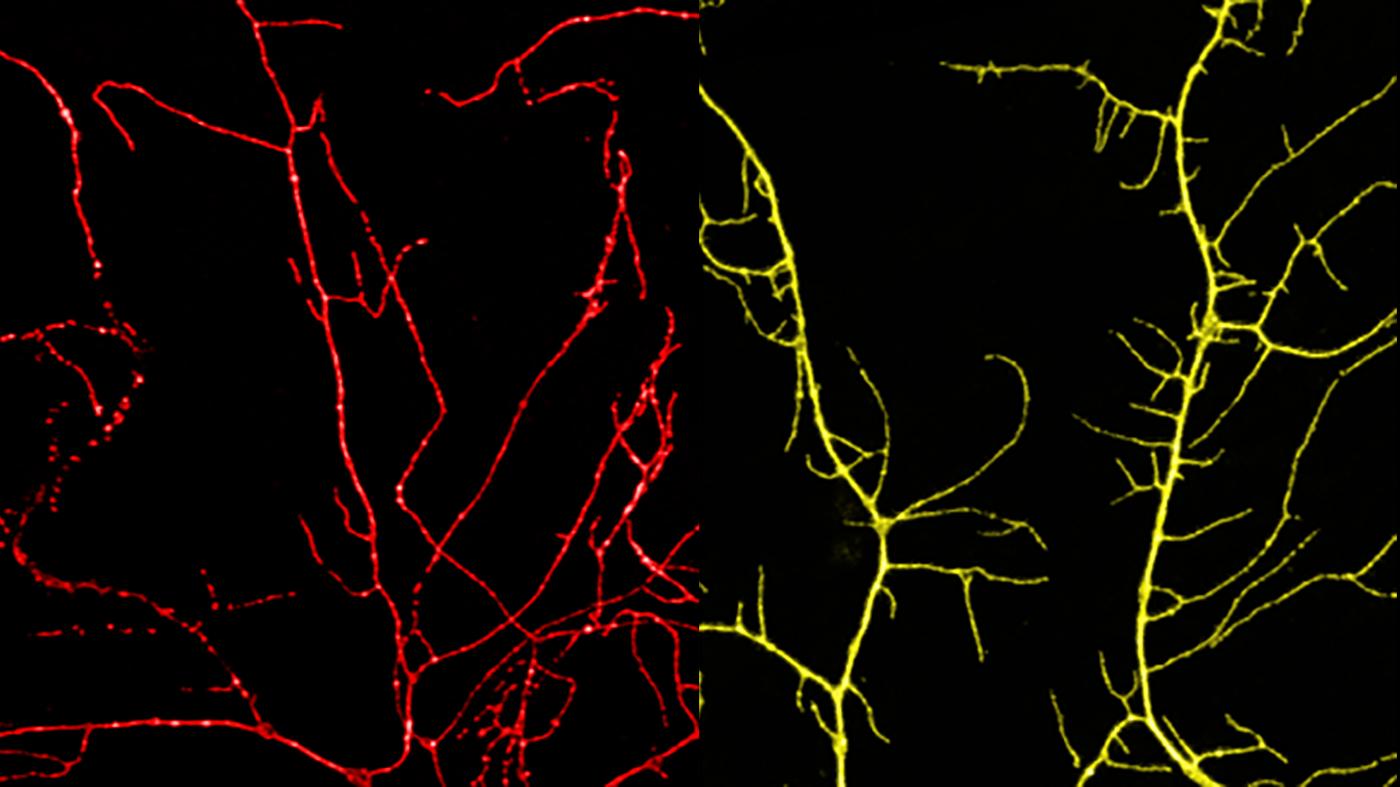
Chemotherapy drugs have been known to cause damage to nerve endings called intraepidermal nerve fibers. Rather than spreading out in an organized fashion, nerve endings exposed to repeated chemotherapy treatments break apart, disappear from their usual places and become disarrayed. Normal integrin production in the body seems to be no match for the severity of this type of nerve-cell damage. Drs. Grueber and Shin hypothesized that boosting integrin levels could help the body overcome this hurdle.
In a series of experiments, first in fruit fly larvae and then in nerve cells from mice, the research team administered the chemotherapy drug paclitaxel. As expected, this drug altered nerve cells’ normal organization; the cells’ branches began to break apart, overlap and clump together.
To test their integrin hypothesis, the researchers artificially increased integrin production in nerve cells in both the fruit fly larvae and mouse cells. They restricted this integrin boost to skin sensory neurons. Next, the researchers administered paclitaxel and monitored for nerve-cell damage.
“To our amazement, this integrin boost proved beneficial to the neurons,” said Dr. Shin. “We observed markedly less clumping or degeneration. The cells remained largely intact, even after being treated by the chemotherapy drug.”
While these results are from animal models, this potential power of preserving the relationship between a cell and its surrounding environment to prevent nerve-cell damage in CIPN shows promise for future studies, the researchers said.
“Further investigations into integrins’ role in nerve-cell repair, as well as a deeper understanding of how nerve-cell branches grow and are maintained, could be key to developing drugs that don’t just blunt symptoms of CIPN — but actually prevent it from arising in the first place,” said Dr. Grueber, who is also a professor of physiology and cellular biophysics and of neuroscience at Columbia’s Vagelos College of Physicians and Surgeons.
Moving forward, the researchers plan to expand their observations in mice and investigate other conditions characterized by neuropathy.
“Diabetes is also known to cause peripheral neuropathy,” said Dr. Shin. “Also, people with diabetes undergoing chemotherapy are at a particularly high risk for CIPN. The mechanisms underlying this connection have not been studied in great detail, but this is something we hope to characterize in detail in the near future.”
###
This paper is titled “Integrins protect sensory neurons in models of paclitaxel-mediated peripheral sensory neuropathy.” Additional contributors include Maria Elena Pero, PhD; Luke A. Hammond; Anita Burgos, PhD; Atul Kumar, PhD; Samantha E. Galindo, PhD; Tanguy Lucas, PhD; and Francesca Bartolini, PhD.
This research was supported by the Thompson Family Foundation Initiative at Columbia University.
The authors declare no competing or financial interests.
Columbia University’s Mortimer B. Zuckerman Mind Brain Behavior Institute brings together a group of world-class scientists and scholars to pursue the most urgent and exciting challenge of our time: understanding the brain and mind. A deeper understanding of the brain promises to transform human health and society. From effective treatments for disorders like Alzheimer’s, Parkinson’s, depression and autism to advances in fields as fundamental as computer science, economics, law, the arts and social policy, the potential for humanity is staggering. To learn more, visit: zuckermaninstitute.columbia.edu.