The developing visual system could easily turn into a traffic pileup. Nerve cells, or neurons, extend roots from the retina, in the back of the eye, several centimeters into the brain. And somehow they seem to know precisely where they are going. At Columbia’s Mortimer B. Zuckerman Mind Brain Behavior Institute, Carol Mason, PhD, studies the circuitry of the visual system to understand how neurons that extend from the eyes make their way to their specified destinations deep in the brain.
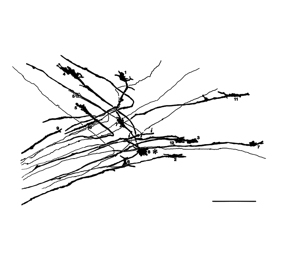
She studies a type of neuron called the retinal ganglion cell (RGC) that stretches from the back of the eye to the thalamus and midbrain, which are sensory relay stations in the middle of the brain. (From these sites, signals are sent to the visual stations in the cortex, the brain region that interprets these signals.) Projecting from one end of each neuron is a thick root called an axon. About half of the RGCs send their axons to the part of the thalamus that sits on the same side of the brain in which they are located, while the axons from the other half of the RGCs cross over to the opposite side. This splitting of the circuit — by which each side of the brain receives signals from both eyes — allows for binocular vision. The question is: How does an axon, once it reaches the intersection at the brain’s midline (called the optic chiasm), know whether to cross over or remain on the same side?
Dr. Mason has studied this question for the past 15 years and has found that the key lies in receptors that are situated on the tips of the growing axons. When one kind of RGC reaches the optic chiasm, molecules there attach to the receptors and block the axon from crossing over, but when another kind arrives, its receptors interact with other molecules and it is allowed to keep going. “It’s like one RGC has the correct subway pass, while the other does not so it can’t get through,” Dr. Mason says. She is now trying to characterize molecular differences between these two RGC types.
All of this work is done with the hope of someday recreating what happened during development, even in the adult.
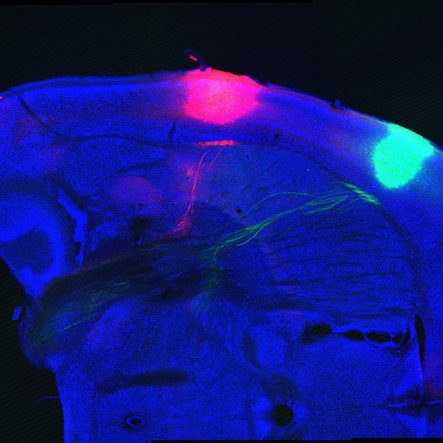
To do this, her lab uses several methods. In one approach, they mutate particular genes in mice and observe how the cells with mutated genes grow. In another, they apply dye or chemical labels to the RGCs and later inspect the brain with a method called clearing. This technique renders the brain transparent except for the highlighted pathways. With high-resolution photography, they can then construct 3D computer renderings of the axons. One important finding from these methods is that the RGCs that cross the optic chiasm arise during an earlier period of gestation, compared to those that do not. “It’s like the oldest born are endowed with the receptors to choose one path, and the youngest born choose a different one,” Dr. Mason says.
Her work also has direct implications for restoring vision. Dr. Mason is looking at how gene activity turns stem cells into RGCs, knowledge that could be used for possible transplantation therapy. Relatedly, she is looking at how to revert mature RGCs to a younger, stem cell–like state so that damaged optic nerves might regrow. “All of this work,” she says, “is done with the hope of someday recreating what happened during development, even in the adult.”